Glaycielli Pereira Santos Mandaro de Assis1,2; Paula Yuri Sacai1,2; Andressa Quércia1; Evelyn Benyhe1; Milena Leal Borges1; Sung Eun Song Watanabe1,2
DOI: 10.17545/eOftalmo/2025.0010
ABSTRACT
Extensive macular atrophy with pseudodrusen is defined as progressive macular degeneration with a broad spectrum of differential diagnoses. As a recently described entity with an unclear pathophysiology, numerous cases are presumed to remain underdiagnosed, suggesting that its prevalence is higher than the current reports in the literature. The present case report describes an 11-year clinical and functional follow-up. The patient was initially diagnosed with age-related macular degeneration. However, with the improved knowledge on extensive macular atrophy with pseudodrusen and the enhanced characterization of clinical and imaging features, the findings proved to be consistent with this entity. Long-term follow-up enabled detailed documentation of the anatomical and functional retinal changes over time.
Keywords: Extensive macular atrophy with pseudodrusen; Multimodal imaging; Retinal electrophysiology; Age-related macular degeneration.
RESUMO
A atrofia macular extensa com pseudodrusas é uma degeneração macular progressiva que apresenta amplo espectro de diagnósticos diferenciais. Por se tratar de uma entidade relativamente recente e com fisiopatologia ainda não completamente elucidada, presume-se que muitos casos permaneçam subdiagnosticados, sugerindo que sua prevalência possa ser superior à atualmente descrita na literatura. O presente relato de caso descreve o acompanhamento clínico e funcional ao longo de 11 anos. Inicialmente, o paciente recebeu o diagnóstico de degeneração macular relacionada à idade. Contudo, com o avanço do conhecimento sobre a atrofia macular extensa com pseudodrusas e sua melhor caracterização clínica e por imagem, o quadro mostrou-se compatível com essa entidade. O seguimento prolongado possibilitou a documentação minuciosa da evolução anatômica e funcional da retina ao longo do tempo.
Palavras-chave: Atrofia macular extensa com pseudodrusas; Análise multimodal; Eletrofisiologia da retina; Degeneração macular relacionada à idade.
INTRODUCTION
Extensive macular atrophy with pseudodrusen (EMAP) is a clinical entity marked by a triad encompassing macular atrophy with predominance along the vertical axis, the presence of pseudodrusen, and peripheral pavingstone-like degeneration, typically bilateral and symmetric1. This progressive condition is associated with cumulative structural and functional retinal impairment over time.
Subsequent studies have demonstrated that this disease may be complicated by the occurrence of subretinal macular neovascularization, an event that potentially contributes to visual deterioration and modifies its clinical presentation2. Since its initial description as a distinct entity3, longitudinal investigations have characterized its progression pattern and functional impact over time4,5.
In clinical practice, diagnosing extensive macular atrophy with pseudodrusen may be challenging, particularly in patients within age ranges similar to those affected by age-related macular degeneration. Studies with long-term follow-up provide a better understanding of the natural history of the disease and its specific structural and functional manifestations.
This case report describes the evolution of progressive chorioretinal atrophy and the long-term clinical and functional follow-up of a patient, highlighting the importance of multimodal analysis in this condition.
CASE REPORT
A 58-year-old man sought ophthalmologic evaluation in 2014 with a complaint of progressive nyctalopia, accompanied by gradual visual acuity (VA) deterioration in both eyes (OU), symmetrically, mainly affecting distance vision. He denied prior ocular disease. His medical history included rheumatic fever at 9 years old, for which he underwent secondary prophylaxis using benzathine penicillin until 18 years old. Furthermore, he reported developing necrotizing fasciitis after appendectomy and being diagnosed with hiatal hernia. No other relevant systemic comorbidities were documented.
Initial examination revealed a VA of 20/16 in the right eye (OD) and 20/32 in the left eye (OS). Examination of the anterior segment and vitreous body revealed no significant changes. Fundus examination revealed pseudodrusen, predominantly between the temporal arcades1 and peripheral pavingstone degeneration (Figure 1). Optical coherence tomography (OCT) showed revealed hyperreflective subretinal drusenoid deposits. Full-field electroretinography (ffERG) showed diffuse retinal dysfunction with predominant rod impairment and reduced cone responses. Multifocal ERG (mfERG) revealed substantial bilateral and symmetric macular cone dysfunction, despite mild asymmetry in VA, indicating that functional impairment preceded structural alterations. Based on this constellation of findings, an initial diagnosis of age-related macular degeneration (AMD) was made.
During follow-up, the patient reported VA fluctuations, worsening nyctalopia, decreased near vision, and photophobia onset. In 2017, pseudodrusen progression became evident, now more diffusely distributed across the posterior pole. Although the disease followed a gradual course, the patient experienced sudden visual decline in the OD in 2018, with VA dropping to 20/200 (OS: 20/40). A subretinal neovascular membrane was identified in the OD and managed with intravitreal anti-VEGF injections.
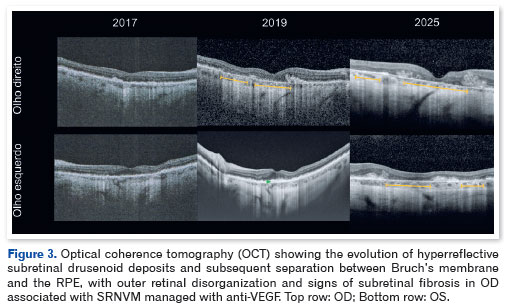
In the 2020 evaluation, central chorioretinal atrophy was documented in the OU, alongside subretinal fibrosis in the OD and centripetal expansion of pavingstone degeneration. Fundus autofluorescence (Figure 2) indicated well-demarcated, symmetric macular hypoautofluorescence with a small central hyperautofluorescent island in the OD and extensive peripheral hyperautofluorescence in the OU, consistent with retinal pigment epithelium (RPE) distress. OCT showed confluent and thickened hyperreflective subretinal drusenoid deposits with progressive separation between Bruch's membrane and RPE.
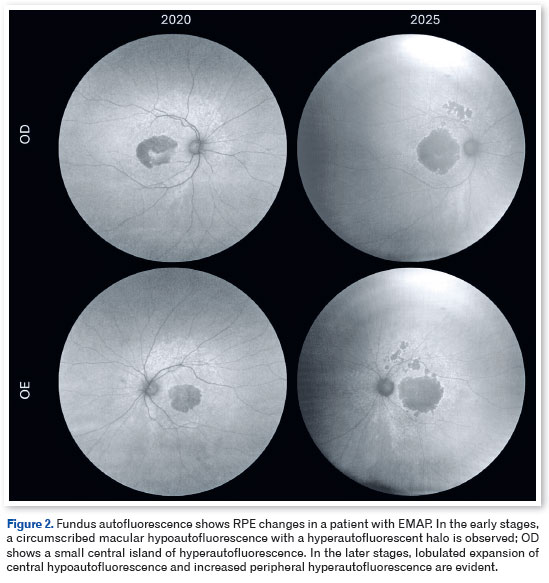
During the most recent evaluation in 2025, the patient reported a marked VA decline—20/320 in the OD and 20/40 in the OS—accompanied by central scotoma, dyschromatopsia, severe photophobia requiring optical filters, reduced contrast sensitivity, and progressive visual-field constriction. Fundus examination revealed the expansion of lobulated chorioretinal atrophy to the arcades and progression of pavingstone degeneration. Autofluorescence imaging showed broad enlargement of the central hypoautofluorescent area and widening of the peripheral hyperautofluorescent rim, highlighting the evolutionary trajectory of the disease. OCT (Figure 3) showed disorganization of the outer retinal layers, alongside disruption of the external limiting membrane, ellipsoid zone, and interdigitation zone, culminating in complete retinal pigment epithelium and outer retinal atrophy.
Electrophysiological testing revealed progressive deterioration of global rod and cone function on ffERG (Figure 4) as well as worsening macular cone function on mfERG (Figure 5).
DISCUSSION
Nyctalopia, photophobia, and dyschromatopsia are distinctive symptoms of extensive macular atrophy with pseudodrusen. The present case was initially interpreted as early age-related macular degeneration4, and vitamin supplementation in accordance with the AREDS 2 protocol was started.
However, the early age at symptom onset, severe nyctalopia, rapid and progressive retinal dysfunction observed on ffERG since symptom onset, evidence of pseudodrusen observed on fundus examination and characterized by OCT5,6, and the discrepancy between symptoms and anatomical findings prompted diagnostic reconsideration. Watanabe et al.7 reported that a prior history of rheumatic fever and the presence of the classic triad of extensive macular atrophy with pseudodrusen reinforced differentiation from AMD.
The present case highlights two relevant aspects of the pathophysiology of extensive macular atrophy with pseudodrusen. The first concern is clinical-functional dissociation: despite prominent nyctalopia and subjective visual complaints, VA remained preserved in the early stages, although ffERG already showed substantial retinal dysfunction. Even in advanced stages, the left eye—which did not develop subretinal macular neovascularization—maintained a VA of 20/40 despite severe electrophysiological impairment, suggesting that VA is not a sensitive marker of disease severity this condition. The second aspect involves function and structure dissociation. Electrophysiological testing conducted in the early stages revealed diffuse retinal dysfunction despite predominantly central anatomical changes, whereas in advanced stages, peripheral areas appeared structurally preserved despite widespread functional loss.
In 2018, the International Society for Clinical Electrophysiology of Vision has standardized photopic negative response, an electrophysiological test aimed at evaluating retinal ganglion cell function8. Watanabe et al. reported that ganglion cell dysfunction may represent one of the earliest electrophysiological findings in extensive macular atrophy with pseudodrusen9, expanding the understanding of disease pathophysiology and suggesting a broader involvement of the inner retinal layers. The incorporation of this protocol may enhance early diagnosis and functional stratification.
In conclusion, the presence of a previous history of rheumatic fever and/or the early and progressive onset of retinal degeneration should raise diagnostic suspicion for EMAP. Multimodal follow-up allowed objective documentation of early structural and functional changes in the retina and macula, demonstrating the progression of retinal involvement to chorioretinal atrophy. The pattern of widespread rod and cone dysfunction observed on full-field ERG, alongside macular cone impairment on mfERG, reinforces the understanding of EMAP as a diffuse retinal disease. These findings highlight the essential role of multimodal evaluation—integrating high-resolution imaging and electrophysiological testing—in the diagnosis, longitudinal monitoring, and appropriate clinical management of patients with EMAP.
REFERENCES
1. Antropoli A, Arrigo A, Bianco L, Berni A, Lamberto F, Saladino A, et al. Quantitative multimodal imaging of extensive macular atrophy with pseudodrusen and geographic atrophy with diffuse trickling pattern. Sci Rep. 2023;13(1):1822.
2. Trinco A, Antropoli A, Bianco L, Zaffalon C, Airaldi M, Lanzani A, et al. Incidence, Characteristics, and Outcomes of Macular Neovascularization in Extensive Macular Atrophy with Pseudodrusen-like Appearance. Retina. 2025;45(9):1689-700.
3. Hamel CP, Meunier I, Arndt C, Salah SB, Lopez S, Bazalgette C, et al. Extensive macular atrophy with pseudodrusen-like appearance: a new clinical entity. Am J Ophthalmol. 2009; 147(4):609-20.
4. Antropoli A, Bianco L, Condroyer C, Antonio A, Navarro J, Dagostinoz D, et al. Extensive macular atrophy with pseudodrusen-like appearance: progression kinetics and late-stage findings. Ophthalmology. 2024;131(10):1175-1184.
5. Romano F, Airaldi M, Cozzi M, Oldani M, Riva E, Bertoni AI, et al. Progression of atrophy and visual outcomes in extensive macular atrophy with pseudodrusen-like appearance. Ophthalmol Sci. 2021;1(1):100016.
6. Romano F, Cozzi M, Salvetti AP. Multimodal imaging of extensive macular atrophy with pseudodrusen-like appearance. Ophthalmol Retina. 2023;7(4):332.
7. Watanabe SES, Quercia AZF, Sacai PY. Electrophysiological findings in extensive macular atrophy with pseudodrusen. Doc Ophthalmol. 2023;147(2):121-130.
8. Frishman L, Sustar M, Kremers J, McAnany JJ, Sarossy M, Tzekov R, et al. ISCEV extended protocol for the photopic negative response (PhNR) of the full-field electroretinogram. Doc Ophthalmol. 2018;136(3):207-211.
9. Watanabe SES, Quercia AZF, de Assis GPSM, Borges ML, Sacai PY. Ganglion cell function loss in extensive macular atrophy with pseudodrusen. Int Ophthalmol. 2025;45(1):338.
AUTHORS INFORMATIONS
Funding: No specific financial support was available for this study.
Conflict of interest: None of the authors have any potential conflict of interest to disclose.
Approved by the following research ethics committee: Universidade Federal de São Paulo (CAAE 73378423.0.0000.5505).
Received on:
January 5, 2026.
Accepted on:
January 9, 2026.