Leonardo Luis Cassoni; Isabela Franco Villela; Francyne Veiga Reis Cyrino
DOI: 10.17545/eOftalmo/2026.v12.005
ABSTRACT
Endophthalmitis is a rare but severe intraocular infection, typically caused by bacterial or fungal pathogens. It may present as an endogenous infection, originating from systemic infections, or an exogenous one, associated with ocular trauma or surgical procedures. Approximately 5%–10% of cases involve endogenous endophthalmitis, which is caused by the hematogenous dissemination of microorganisms that cross the blood–ocular barrier. Immunocompromised individuals are at increased risk of endogenous endophthalmitis attributed to the translocation of microorganisms from their own microbiota. Herein, we report a patient who developed endogenous endophthalmitis during social isolation in the coronavirus disease 2019 (COVID-19) pandemic, after severe acute respiratory syndrome coronavirus 2 infection. This report aimed to describe the clinical presentation, management, and anatomical and visual outcomes of endogenous endophthalmitis, and to discuss potential associations between ocular infections and COVID-19, without implying direct causality.
Keywords: Endogenous endophthalmitis; Staphylococcus hominis; COVID-19; Case report; Eye infection.
RESUMO
A endoftalmite é uma infecção intraocular rara e grave, geralmente causada por patógenos bacterianos ou fúngicos. Pode apresentar-se de forma endógena, originada de infecções sistêmicas, ou exógena, associada a trauma ou procedimentos cirúrgicos oculares. Cerca de 5% a 10% dos casos de endoftalmite são endógenos, resultantes da disseminação hematogênica de microrganismos que ultrapassam a barreira hemato-ocular. Indivíduos imunocomprometidos apresentam risco aumentado de endoftalmite endógena, frequentemente relacionada à translocação de microrganismos da própria microbiota. Este relato descreve um caso de endoftalmite endógena ocorrido durante o isolamento social da pandemia de COVID-19, após infecção por SARS-CoV-2. O objetivo é apresentar a manifestação clínica, o manejo terapêutico e os desfechos anatômicos e visuais, além de discutir possíveis associações entre infecções oculares e a COVID-19, sem estabelecer relação causal direta.
Palavras-chave: Endoftalmite endógena; Staphylococcus hominis; COVID-19; Relato de caso; Infecção ocular.
INTRODUCTION
Endophthalmitis is a rare but potentially severe infection involving the internal structures of the eye, usually caused by bacteria or fungi. It may be classified as endogenous, originating from systemic infections, or exogenous, caused by external inoculation after ocular trauma or surgical procedures.
Approximately 5%–10% of cases involve endogenous endophthalmitis, occurring when microorganisms from distant infectious foci enter the bloodstream and subsequently cross the blood–ocular barrier1–4. The most common primary sources of infection include the urinary tract, hepatobiliary system, and intravascular catheters1,4. In particular, immunocompromised individuals are susceptible, as endogenous infection may be attributed to the translocation of microorganisms from their own microbiota.
Herein, we describe a patient diagnosed with endogenous endophthalmitis during social isolation in the coronavirus disease 2019 (COVID-19) pandemic, after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. This case report aimed to present the clinical features, management, and anatomical and visual outcomes of endogenous endophthalmitis, and to discuss possible associations between ocular infections and COVID-19 in a cautious and hypothesis-driven manner.
CASE REPORT
A 65-year-old male patient was referred to our service due to blurred vision in his right eye that progressed over a 2-month period. The condition was painless, and it occurred without preceding trauma or previous ocular surgery. He reported recent hospitalization due to COVID-19, during which he developed cholelithiasis and underwent cholecystectomy, in addition to treatment for a urinary tract infection. Fifteen days after hospital discharge, he noticed the onset of floaters in his right eye.
The patient sought ophthalmologic evaluation, which revealed anterior chamber inflammation and vitritis. Treatment with oral sulfamethoxazole–trimethoprim (800mg/160 mg twice daily), topical prednisolone acetate 1%, and tropicamide 1% was initiated and maintained for approximately 2 months. However, visual improvement was not observed. The patient was then referred to our service for further examination. His medical history was significant for systemic arterial hypertension and diabetes mellitus.
Based on ocular examination, the patient's visual acuity in the right eye was limited to light perception. There was ciliary injection, an anterior chamber reaction (graded as 2+ with fibrin), 360º posterior synechiae, and nuclear cataract (graded as 2+). Visualization of the posterior segment was not possible due to dense vitreous opacity. The left eye had a visual acuity of 20/20, without anterior or posterior segment abnormalities. The intraocular pressure values were 11 mmHg in the right eye and 12 mmHg in the left eye.
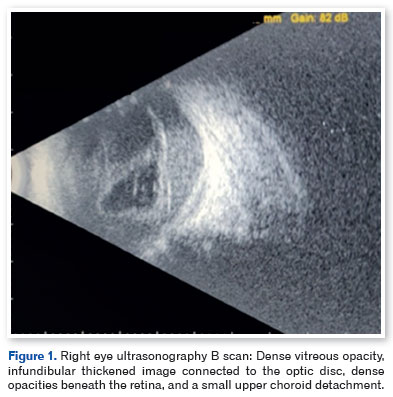
B-scan ultrasonography revealed dense vitreous opacity, a thickened infundibular-shaped structure connected to the optic disc, dense subretinal opacities, and a small superior choroidal detachment (Figure 1). Paracentesis of the aqueous humor was performed. Polymerase chain reaction (PCR) yielded negative results for Toxoplasma gondii, Cytomegalovirus, herpes simplex virus type 1, and varicella-zoster virus. At this stage, the working diagnosis was panuveitis of unknown etiology, and diagnostic vitrectomy was indicated.
The surgery comprised vitreous aspiration for bacterial and fungal cultures via pars plana, followed by synechiolysis, pars plana vitrectomy, endolaser photocoagulation, and silicone oil injection. Intravitreal antibiotics (including ceftazidime, amphotericin B, and vancomycin) were administered. Vitreous samples were sent for culture, antimicrobial susceptibility testing, and PCR.
Intraoperatively, dense vitritis, total retinal detachment, and dense subretinal fluid were observed. A retinal fragment was collected for anatomopathological analysis. Vitreous PCR yielded negative results for the tested pathogens. However, the culture yielded growth of Staphylococcus hominis subsp. hominis, which confirmed the diagnosis of endogenous endophthalmitis. Despite surgical intervention and intravitreal antibiotic therapy, the patient progressed with severe visual loss.
DISCUSSION
Coagulase-negative staphylococci (CoNS), such as S. epidermidis and S. saprophyticus, are common constituents of the ocular surface microbiota. Further, they are the most frequent etiologic agents of post-procedural endophthalmitis2. S. hominis, also a member of the CoNS group, is part of the normal skin and mucosal microbiota and may be more prevalent than S. epidermidis in healthy individuals. However, it remains a rare cause of endophthalmitis3–7.
The pathogenic potential of CoNS is closely related to their ability to form biofilms, which facilitate adhesion to both biotic surfaces, such as the conjunctiva, and abiotic materials, including medical devices and catheters8. Patients with diabetes mellitus exhibit increased conjunctival biofilm formation, which may contribute to greater susceptibility to infection7–9. In addition, CoNS infections are more frequently reported in immunocompromised individuals and those with indwelling foreign bodies or systemic comorbidities5,6.
During the COVID-19 pandemic, several reports have described cases of endogenous endophthalmitis occurring in temporal association with SARS-CoV-2 infection. These cases have been observed both during hospitalization and in the postinfectious period, often in patients with systemic comorbidities, recent infections, or immunological imbalance10. Importantly, these findings support an association rather than a direct causal relationship.
Beyond direct viral effects, the pandemic context itself is associated with factors, including reduced physical activity, delayed access to specialized medical care, and inappropriate antimicrobial use, that may indirectly increase susceptibility to secondary infections 11–13. These factors may contribute to systemic vulnerability, particularly in patients with chronic diseases.
From a pathophysiological perspective, several organs—such as the kidneys and gastrointestinal tract—express higher levels of angiotensin-converting enzyme 2, the main cellular entry receptor for SARS-CoV-214,15. Viral infection of these organs may result in transient or persistent dysfunction, potentially contributing to immune dysregulation during the recovery phase16. Such post-viral immune imbalance has been hypothesized as a possible predisposing factor for opportunistic infections, including endogenous endophthalmitis. Nonetheless, this mechanism remains speculative.
In the current case, the patient developed endogenous endophthalmitis after COVID-19 infection, urinary tract infection, and cholecystitis that required cholecystectomy. A postinfectious immune imbalance related to SARS-CoV-2 may be a contributing factor. Nevertheless, this hypothesis should be interpreted with caution. Diabetes mellitus alone is a well-established risk factor for CoNS infections and may independently explain the occurrence of endogenous endophthalmitis in this patient8,17. Nevertheless, the temporal association with COVID-19 and subsequent systemic complications underscores the importance of heightened clinical vigilance in vulnerable individuals.
In conclusion, social isolation is essential for decreasing the spread of COVID-19. However, it is accompanied by unintended consequences, including reduced physical activity, delays in medical care, and changes in host–microbiota balance11–13. In addition, COVID-19 is associated with systemic effects and transient immune dysregulation, which may increase susceptibility to secondary infections, particularly in immunocompromised patients14–16.
The current case emphasizes the importance of considering opportunistic pathogens such as S. hominis in the differential diagnosis of endogenous endophthalmitis, particularly in patients with multiple systemic risk factors. Further, this case highlights the importance of considering opportunistic pathogens such as S. hominis in endogenous endophthalmitis, particularly in immunocompromised patients. Although a direct causal relationship with SARS-CoV-2 infection cannot be established, the temporal association and systemic complications observed should be further investigated.
AUTHORS' CONTRIBUTIONS:
Significant contribution to conception and design: Leonardo Luis Cassoni. Data Acquisition: Leonardo Luis Cassoni. Data Analysis and interpretation: Leonardo Luis Cassoni. Manuscript Drafting: Leonardo Luis Cassoni, Isabela Franco Villela. Significant intellectual content revision of the manuscript: Francyne Veiga Reis Cyrino, Isabela Franco Villela. Final approval of the submitted manuscript: Leonardo Luis Cassoni, Francyne Veiga Reis Cyrino, Isabela Franco Villela. Statistical analysis: Leonardo Luis Cassoni.Obtaining funding: not applicable. Supervision of Administrative, technical, or material support: Francyne Veiga Reis Cyrino. Research group leadership: Francyne Veiga Reis Cyrino.
REFERENCES
1. Bohigian GM, Olk RJ. Factors associated with a poor visual result in endophthalmitis. Am J Ophthalmol 1986;101(3):332-41.
2. Puliafito CA, Baker AS, Haaf J, Foster CS. Infectious endophthalmitis. Review of 36 cases. Ophthalmology. 1982;89(8):921-929.
3. Shrader SK, Band JD, Lauter CB, Murphy P. The clinical spectrum of endophthalmitis: incidence, predisposing factors, and features influencing outcome. J Infect Dis 1990;162(1):115-120.
4. Jackson TL, Paraskevopoulos T, Georgalas I. Systematic Review Of 342 Cases Of Endogenous Bacterial Endophthalmitis. Surv Ophthalmol. 2014;59(6):627-35.
5. Piette A, Verschraegen G. Role of coagulase-negative staphylococci in human disease. Vet Microbiol. 2009;134(1-2):45-54.
6. Yu W, Kim HK, Rauch S, Schneewind O, Missiakas D. Pathogenic conversion of coagulase-negative staphylococci. Microbes Infect. 2017;19(2):101-109.
7. Gautam V, Sethuraman N, Kaur R, Sachdev S, Marwaha N, Ray P. Changing epidemiology of coagulase-negative staphylococci in normal flora of skin. Indian J Med Microbiol. 2017;35(2):277-278.
8. Kıvanç SA, Arık G, Akova-Budak B, Kıvanç M. Biofilm forming capacity and antibiotic susceptibility of Staphylococcus spp. with the icaA/icaD/bap genotype isolated from ocular surface of patients with diabetes. Malawi Med J. 2018;30(4):243-9.
9. Natsis, NE, Cohen, PR. Coagulase-Negative Staphylococcus Skin and Soft Tissue Infections. Am J Clin Dermatol. 2018;19(5):671-677.
10. Verma S, Walinjkar JA. A case of endogenous endophthalmitis in COVID 19 pandemic. Apollo Med. 2020;17(3):229-32.
11. Goethals L, Barth N, Guyot J, Hupin D, Celarier T, Bongue B. Impact of home quarantine on physical activity among older adults living at home during the COVID-19 pandemic: Qualitative interview study. J Med Internet Res. 2020;22(5):1-5.
12. Wallis CJD, Catto JWF, Finelli A, Glaser AW, Gore JL, Loeb S, et al. The Impact of the COVID-19 Pandemic on Genitourinary Cancer Care: Re-envisioning the Future. Eur Urol. 2020;78(5):731-742.
13. Zhang A, Hobman EV, De Barro P, Young A, Carter DJ, Byrne M. Self-Medication with Antibiotics for Protection against COVID-19: The Role of Psychological Distress, Knowledge of, and Experiences with Antibiotics. Antibiotics (Basel). 2021;10(3):232.
14. Zou X, Chen K, Zou J, Han P, Hao J, Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020;14(2):185-192.
15. Lin L, Jiang X, Zhang Z, Huang S, Zhang Z, Fang Z, et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut. 2020;69(6):997-1001.
16. Balachandar V, Mahalaxmi I, Subramaniam M, Kaavya J, Senthil Kumar N, Laldinmawii G, et al. Follow-up studies in COVID-19 recovered patients - is it mandatory? Sci Total Environ. 2020 Aug 10;729:139021.
17. Armstrong DG, Lanthier J, Lelievre P, Edelson GW. Methicillin-resistant coagulase-negative staphylococcal osteomyelitis and its relationship to broad-spectrum oral antibiosis in a predominantly diabetic population. J Foot Ankle Surg. 1995;34(6):563-6.
| AUTHO'S INFORMATION |
|
 |
» Leonardo Luis Cassoni https://orcid.org/0000-0003-1427-9105 http://lattes.cnpq.br/4681844858936626 |
 |
» Francyne Veiga Reis Cyrino https://orcid.org/0000-0002-1892-6210 http://lattes.cnpq.br/0720130883701897 |
 |
» Isabela Franco Villela https://orcid.org/0000-0002-1736-6717 http://lattes.cnpq.br/7786081137581336 |
Funding: No specific financial support was available for this study.
Approved by the following research ethics committee: Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto – USP – HCFMRP/USP (CAAE: 91209825.6.0000.5440).
Conflict of interest: None of the authors have any potential conflict of interest to disclose.
Received on:
January 10, 2026.
Accepted on:
February 11, 2026.