Marcia Brazuna1; Isabella Felix Q. Yamane2; Matheus A. P. Vieira1
DOI: 10.17545/eOftalmo/2025.0014
ABSTRACT
Extensive macular atrophy with pseudodrusen-like appearance is a rare, progressive maculopathy characterized by bilateral, symmetrical outer retinal atrophy, associated with pseudodrusen deposits and significant visual impairment. We report the case of a 56-year-old patient diagnosed with extensive macular atrophy with pseudodrusen-like appearance who had a history of childhood poststreptococcal glomerulonephritis and long-term exposure to benzathine penicillin. The patient presented with progressive visual acuity decline and nyctalopia over 5 yr, with a final best-corrected visual acuity of 20/30 in the right eye and 20/400 in the left eye. Optical coherence tomography demonstrated pseudodrusen deposits predominantly in the posterior pole as well as focal separation between the retinal pigment epithelium and Bruch's membrane. Fundus autofluorescence revealed sharply demarcated hypoautofluorescent areas, and full-field electroretinography showed bilateral cone-predominant dysfunction, the most frequently reported electrophysiologic pattern in extensive macular atrophy with pseudodrusen-like appearance. Serum complement C3 and C4 levels and C-reactive protein remained within normal limits, with no evidence of active systemic inflammation. To the best of our knowledge, this is the first report describing extensive macular atrophy with pseudodrusen-like appearance in a patient with a history of poststreptococcal glomerulonephritis, suggesting a possible immunoinflammatory association; however, current evidence does not support a causal relationship.
Keywords: EMAP; Pseudodrusen; Complement; glomerulonephritis; Benzathine penicillin
RESUMO
A atrofia macular extensa com pseudodrusas (EMAP) é uma maculopatia rara e progressiva, caracterizada por atrofia bilateral e simétrica de retina externa associada a depósitos tipo pseudodrusas e perda visual acentuada. Relata-se o caso de um paciente de 56 anos com diagnóstico de atrofia macular extensa com pseudodrusas, antecedente de glomerulonefrite pós-estreptocócica na infância e uso prolongado de penicilina benzatina. O paciente apresentava piora progressiva da acuidade visual e nictalopia há cinco anos, com acuidade final de 20/30 no olho direito e 20/400 no esquerdo. A tomografia de coerência óptica revelou pseudodrusas mais evidentes no polo posterior e separação focal entre o epitélio pigmentar da retina e a membrana de Bruch. A autofluorescência mostrou zonas hipoautofluorescentes bem delimitadas, e o eletrorretinograma evidenciou disfunção retiniana bilateral com predomínio de comprometimento cone-mediado, padrão descrito como o mais frequente na atrofia macular extensa com pseudodrusas. Os níveis séricos de C3, C4 e PCR estavam normais, sem sinais de inflamação sistêmica. Até onde sabemos, não há relatos prévios de EMAP em paciente com antecedente de glomerulonefrite pós-estreptocócica, achado que reforça a possibilidade de mecanismo imunoinflamatório da doença, embora sem relação causal demonstrada.
Palavras-chave: EMAP; Pseudodrusas; Complemento; Glomerulonefrite; Penicilina benzatina.
INTRODUCTION
Extensive macular atrophy with pseudodrusen-like appearance (EMAP) is a rare maculopathy characterized by bilateral, symmetrical, and rapidly progressive chorioretinal atrophy, with visual loss typically manifesting in the fifth to sixth decades of life1,2. Clinically, patients present with nyctalopia, central and peripheral scotomas, and a progressive decline in visual acuity. Funduscopic examination usually reveals vertically oriented macular involvement with pseudodrusen deposits extending into the midperiphery3-5.
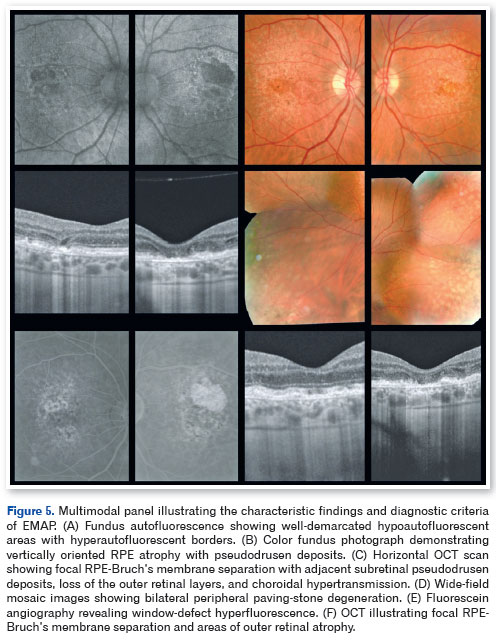
Although EMAP presents a distinct phenotype, it is essential to differentiate it from the diffuse-trickling form of geographic atrophy associated with age-related macular degeneration, which is known for a more aggressive clinical course5. To facilitate this distinction, Antropoli et al.3 proposed diagnostic criteria that include bilateral and symmetrical atrophy with multilobulated, vertically oriented borders; diffuse pseudodrusen deposits; peripheral paving-stone degeneration; and characteristic findings on multimodal imaging. Fundus autofluorescence typically demonstrates sharply demarcated hypoautofluorescent areas with isoautofluorescent to hyperautofluorescent borders, while optical coherence tomography (OCT) reveals loss of the outer retinal layers, alterations in the retinal pigment epithelium (RPE)-Bruch's membrane complex, and choroidal thinning3,4,6. Electrophysiologic abnormalities, predominantly cone-mediated dysfunction, further support the functional diagnosis7,8.
Beyond structural and functional findings, environmental and immunologic factors have been implicated as potential risk cofactors, including occupational exposures and persistent activation of the complement cascade, suggesting an immunoinflammatory pathogenesis modulated by epigenetic mechanisms9-15. Investigation of associated systemic factors remains relevant, particularly in light of emerging evidence of chronic immune activation involving complement pathways, underscoring the importance of contextualizing cases with inflammatory or infectious systemic antecedents.
In Brazil, Moreira-Neto et al.16 reported a high prevalence of rheumatic fever (90.9%) and long-term benzathine penicillin use (94.8%) among patients with EMAP, suggesting that immune activation mediated by streptococcal infection may contribute to disease development. Interestingly, they also observed that antibiotic use was associated with reduced atrophic extent, suggesting a potential modulatory or protective effect. The present report describes a case of EMAP in a patient with a history of poststreptococcal glomerulonephritis and prolonged benzathine penicillin exposure—an association not previously identified in the literature.
This case expands the spectrum of immune-mediated systemic factors potentially linked to EMAP and highlights the importance of evaluating immunologic antecedents in patients presenting with compatible multimodal imaging features.
CASE REPORT
A 56-year-old male was referred for specialized ophthalmologic evaluation due to nyctalopia and progressive, bilateral decline in visual acuity over the preceding 5 yr, which was more pronounced under low-light conditions. He denied ocular pain, metamorphopsia, or prior use of retinotoxic medications. He regularly wore corrective lenses for myopia and astigmatism.
His medical history included systemic arterial hypertension, type 2 diabetes mellitus, and a previous acute myocardial infarction, all of which were well controlled. During childhood, he experienced poststreptococcal glomerulonephritis, which was treated with multiple injections of benzathine penicillin. He also reported intermittent and prolonged antibiotic use throughout adulthood, although detailed documentation regarding indications was unavailable. There was no family history of hereditary retinal or autoimmune ocular disease.
Best-corrected visual acuity was 20/30 in the right eye and 20/400 in the left eye. Slit-lamp biomicroscopy revealed nuclear cataract (N2) in both eyes. Fundus examination demonstrated bilateral macular atrophy with a vertically oriented pattern, pigmentary irregularities, and yellowish pseudodrusen deposits predominantly in the posterior pole, accompanied by peripheral paving-stone degeneration (Figures 1 and 2).
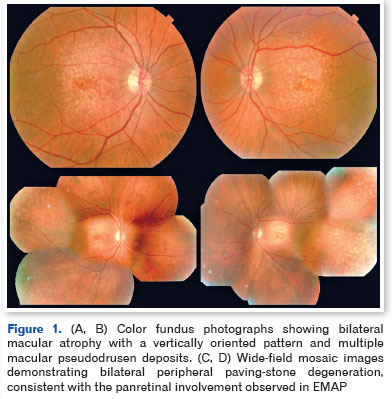
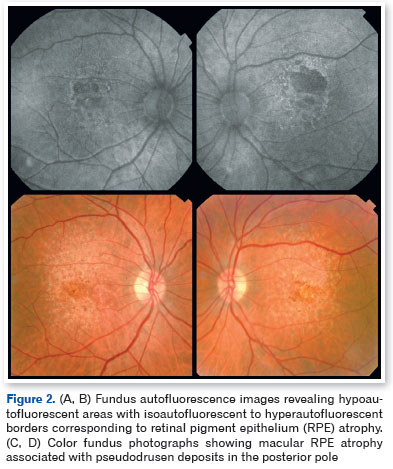
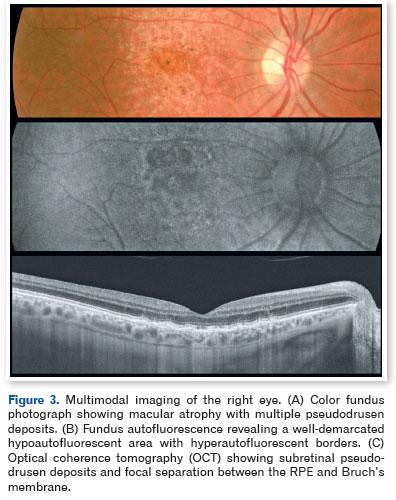
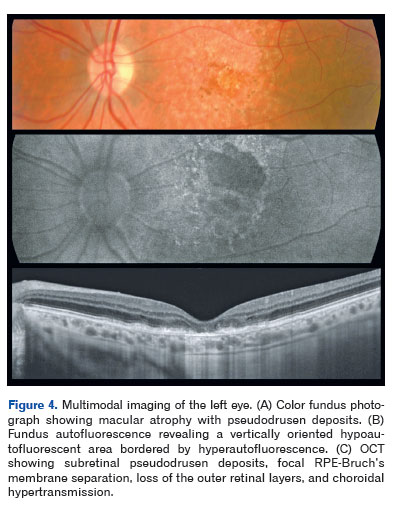
Optical coherence tomography demonstrated pseudodrusen deposits at the posterior pole, focal separation between the RPE and Bruch's membrane, and areas of macular atrophy. Fundus autofluorescence revealed sharply demarcated hypoautofluorescent zones with isoautofluorescent to hyperautofluorescent borders, consistent with RPE loss (Figures 3-5).
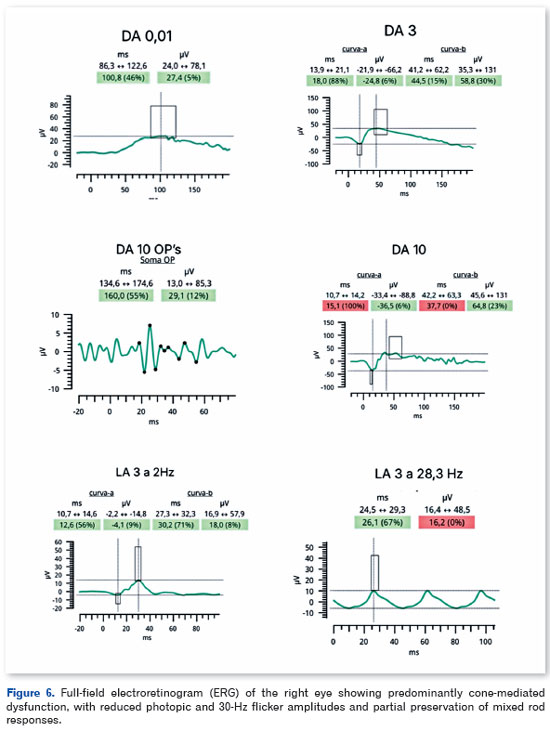
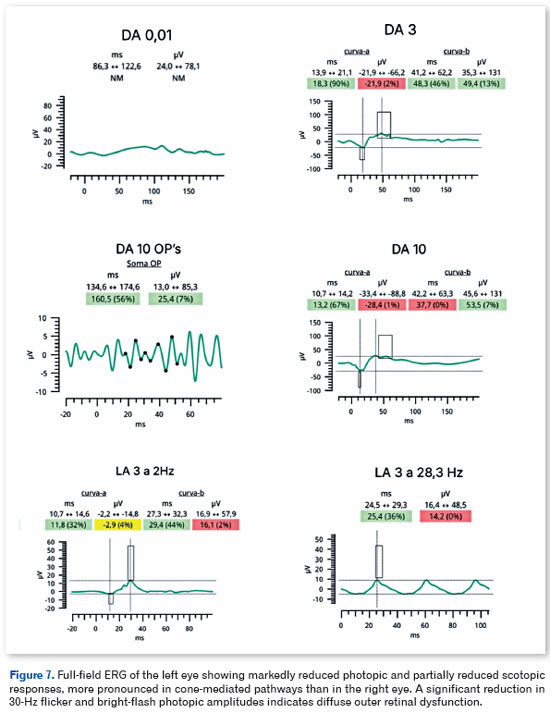
Full-field electroretinography, performed in accordance with International Society for Clinical Electrophysiology of Vision (ISCEV) standards, demonstrated bilateral, predominantly cone-mediated retinal dysfunction, characterized by reduced photopic and 30-Hz flicker amplitudes with partial preservation of mixed rod responses, more pronounced in the left eye (Figures 6 and 7).
Laboratory evaluation revealed normal serum complement levels (C3: 105mg/dL; C4: 28.6mg/dL) and inflammatory markers within normal limits (C-reactive protein [CRP]: 0.1mg/dL; erythrocyte sedimentation rate: 13mm/h), arguing against active systemic inflammation. Hematologic and cardiovascular assessments showed no relevant abnormalities.
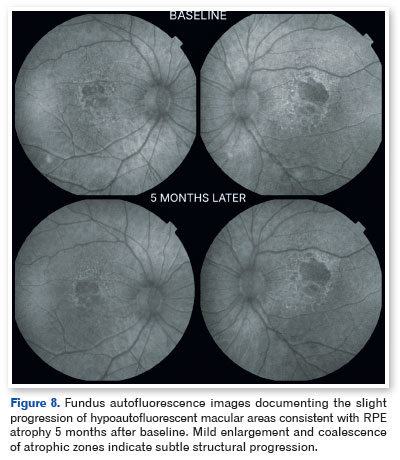
Based on the clinical presentation and multimodal imaging findings, a diagnosis of EMAP was established. The patient was counseled regarding the degenerative nature of the disease and the importance of regular follow-up. At 5-month follow-up, slight bilateral progression of retinal atrophy was observed, accompanied by mild functional deterioration of vision in the left eye (Figures 8 and 9).
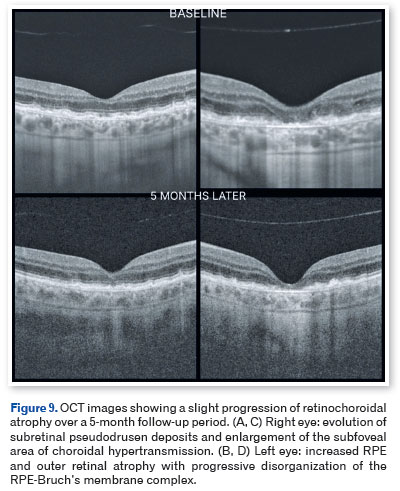
DISCUSSION
The present case fulfills the clinical and multimodal imaging criteria described for EMAP, including extensive bilateral, vertically oriented macular atrophy, pseudodrusen deposits, and peripheral paving-stone degeneration1-4,6. Since its initial description by Hamel et al.1, EMAP has been recognized as a degenerative entity distinct from atrophic age-related macular degeneration due to its earlier age at onset and more rapid progression with foveal involvement2-4,17. The structural findings observed in this case further reinforce the panretinal character of the disease.
Full-field ERG demonstrated bilateral, predominantly cone-mediated retinal dysfunction, characterized by diffuse reductions in photopic and flicker amplitudes with partial preservation of mixed rod responses. These findings support the presence of diffuse outer retinal dysfunction and are consistent with the neurodegenerative profile of EMAP7,8.
The pathophysiology of EMAP remains incompletely understood; however, accumulating evidence suggests that abnormal and sustained activation of the complement cascade plays a central role10,12,13. This chronic, low-grade inflammatory activation leads to progressive loss of the outer retinal layers and subsequent retinochoroidal atrophy. The underlying mechanisms appear to be modulated by a combination of genetic susceptibility and environmental factors11,13,14. Notably, the absence of systemic alterations in complement components C3 and C4 or CRP, as observed in this patient, does not preclude complement involvement, as activation is thought to occur predominantly at the local tissue level. Recent studies further suggest that epigenetic regulation of complement pathways and inflammatory responses may contribute to the phenotypic variability of EMAP10-13.
Molecular mimicry involving streptococcal antigens and retinal proteins has been proposed as a potential biological mechanism linking prior streptococcal infections to immune-mediated retinal degeneration. This hypothesis supports the concept of persistent subclinical inflammation, chronic complement activation, and cumulative damage to retinochoroidal structures18,19.
In a Brazilian cohort, Moreira-Neto et al.16 reported a high prevalence of rheumatic fever and prolonged benzathine penicillin use among patients with EMAP, suggesting that poststreptococcal immune mechanisms and chronic immunomodulation related to treatment may act as triggers in predisposed individuals. Similarly, Watanabe et al.7 demonstrated diffuse cone-mediated dysfunction, corroborating the extent of functional impairment typically observed in EMAP.
Unlike glomerulopathies driven by alternative complement pathway dysregulation—such as C3 glomerulopathy and dense-deposit disease, in which primary complement dysfunction has been more directly linked to retinal alterations—poststreptococcal glomerulonephritis is classically mediated by immune complex deposition.10,12,13,18.
The association observed in this case between poststreptococcal glomerulonephritis and EMAP suggests a potential shared immunologic vulnerability, although no causal relationship can be established. This report expands the spectrum of systemic conditions potentially associated with EMAP and reinforces the plausibility of a chronic autoimmune response acting as a common axis linking systemic immune phenomena to the ocular manifestations of the disease.
REFERENCES
1. Hamel CP, Meunier I, Arndt C, Ben Salah S, Lopez S, Bazalgette C, et al. Extensive macular atrophy with pseudodrusen-like appearance: a new clinical entity. Am J Ophthalmol. 2009;147(4): 609-20.
2. Carla MM, Giannuzzi F, Boselli F, Crincoli E, Rizzo S. Extensive macular atrophy with pseudodrusen-like appearance: comprehensive review of the literature. Graefes Arch Clin Exp Ophthalmol. 2024;262(10):3085-97.
3. Antropoli A, Bianco L, Romano F, Trinco A, Arrigo A, Benadji A, et al. Extensive macular atrophy with pseudodrusen-like appearance (EMAP): clinical characteristics, diagnostic criteria, and insights from allied inherited retinal diseases and age-related macular degeneration. Prog Retin Eye Res. 2025;104:101320.
4. Antropoli A, Bianco L, Condroyer C, Antonio A, Navarro J, D'Agostino D, et al. Extensive macular atrophy with pseudodrusen-like appearance: progression kinetics and late-stage findings. Ophthalmology. 2024;131(10):1175-84.
5. Antropoli A, Arrigo A, Bianco L, Berni A, Lamberto F, Saladino A, et al. Quantitative multimodal imaging of extensive macular atrophy with pseudodrusen and geographic atrophy with diffuse trickling pattern. Sci Rep. 2023;13(1):1822.
6. Battaglia Parodi M, Antropoli A, Bianco L, Arrigo A, Pili L, Saladino A, et al. Peripheral retinal involvement in extensive macular atrophy with pseudodrusen-like deposits. Ophthalmol Retina. 2023;7(10):910-7.
7. Watanabe SES, Quercia AZF, Sacai PY. Electrophysiological findings in extensive macular atrophy with pseudodrusen. Doc Ophthalmol. 2023;147(2):121-30.
8. Robson AG, Frishman LJ, Grigg J, Hamilton R, Jeffrey BG, Kondo M, et al. ISCEV Standard for full-field clinical electroretinography (2022 update). Doc Ophthalmol. 2022;144(3):165-77.
9. Spaide RF, Vavvas DG. Complement inhibition for geographic atrophy: review of salient functional outcomes and perspective. Retina. 2023;43(7):1064-9.
10. Morgan BP, Harris CL. Complement, a target for therapy in inflammatory and degenerative diseases. Nat Rev Drug Discov. 2015;14(12):857-77.
11. Lambert NG, ElShelmani H, Singh MK, Mansergh FC, Wride MA, Padilla M, et al. Risk factors and biomarkers of age-related macular degeneration. Prog Retin Eye Res. 2016;54:64-102.
12. Armento A, Ueffing M, Clark SJ. The complement system in age-related macular degeneration. Cell Mol Life Sci. 2021;78(10): 4487-505.
13. Adamus G. Importance of autoimmune responses in progression of retinal degeneration initiated by gene mutations. Front Med (Lausanne). 2021;8:672444.
14. Douillard A, Picot MC, Delcourt C, Defoort-Dhellemmes S, Marzouka NA, Lacroux A, et al. Dietary, environmental, and genetic risk factors of extensive macular atrophy with pseudodrusen: a severe bilateral macular atrophy of middle-aged patients. Sci Rep. 2018;8(1):6840.
15. Douillard A, Picot MC, Delcourt C, Lacroux A, Zanlonghi X, Puech B, et al. Clinical characteristics and risk factors of extensive macular atrophy with pseudodrusen: the EMAP case-control national clinical trial. Ophthalmology. 2016;123(9):1865-73.
16. Moreira-Neto CA, Schmidt Andujar RA, Chao JCT, Vasconcelos H, Alves FEE, Rodrigues GD, et al. Rheumatic fever and long-term use of benzathine penicillin as possible risk factors for extensive macular atrophy with pseudodrusen in a Brazilian cohort. Int J Retina Vitreous. 2024;10(1):75.
17. Romano F, Cozzi M, Monteduro D, Oldani M, Boon CJF, Staurenghi G, et al. Natural course and classification of extensive macular atrophy with pseudodrusen-like appearance. Retina. 2023;43(3):402-11.
18. Cunningham MW. Molecular mimicry, autoimmunity, and infection: the cross-reactive antigens of group A streptococci and their sequelae. Microbiol Spectr. 2019;7(4).
19. Lerner MP, Donoso LA, Nordquist RE, Cunningham MW. Immunological mimicry between retinal S-antigen and group A streptococcal M proteins. Autoimmunity. 1995;22(2):95-106.
| AUTHORS INFORMATIONS |
|
 |
» Marcia Brazuna https://orcid.org/0009-0008-0688-8596 http://lattes.cnpq.br/9692116316379230 |
 |
» Matheus A. P. Vieira https://orcid.org/0009-0002-8178-7978 http://lattes.cnpq.br/3913686891761563 |
 |
» Isabella Felix Q. Yamane https://orcid.org/0009-0001-3418-8596 http://lattes.cnpq.br/0474957487960468 |
Funding: No specific financial support was available for this study.
Conflict of interest: None of the authors have any potential conflict of interest to disclose.
Received on:
January 10, 2025.
Accepted on:
September 24, 2025.