Luiz Guilherme de Moraes Matheus1; Rafael Nazaro Cassar Camargo2; Bernardo Kaplan Moscovici3; Ulysses Tachibana4
DOI: 10.17545/e-oftalmo.cbo/2017.86
ABSTRACT
The conjunctiva may be the site of innumerable lesions, among them tumors. The conjunctival tumors may present in innumerablehistological forms, with variable characteristics. Early diagnosis and appropriate treatment are extremely important for patient healing. This article aims to present the types of conjunctival tumors of epithelial origin, as well as their diagnosis and therapeutic possibilities.
Keywords: Ophthalmology; Conjunctival Diseases; Conjunctival Neoplasms;
RESUMO
A conjuntiva pode ser sede de inúmeras lesões, dentre elas os tumores. Os tumores conjuntivais podem se apresentar de inúmeras formas histológicas, com características variáveis. O diagnóstico precoce e o adequado tratamento são de extrema importância para a cura do paciente. O presente artigo apresenta os tipos de tumores de conjuntiva de origem epitelial, bem como seu diagnóstico e possibilidades terapêuticas.
Palavras-chave: Oftalmologia; Doenças da Túnica Conjuntiva; Neoplasias da Túnica Conjuntiva;
INTRODUCTION
The conjunctiva is an accessory structure of the eye, which is formed by a mucous membrane that is characterized by a posterior lining of the eyelids and anterior covering of the surface of the eye. The conjunctiva that lines the eyelids is referred to as the palpebral conjunctiva, and the conjunctiva that covers the surface of the eye to the edge of the cornea is known as the bulbar conjunctiva. This structure is tightly bound to the eyelid and loosely bound to the eyeball. The latter junction forms the fornix, which is a cul-de-sac formed in the superior and inferior bands between the eyeball and eyelids.1 Histologically, the conjunctiva has prismatic stratified epithelium, and its lamina propria consists of loose connective tissue.2 The conjunctiva may be compromised by many different lesions of diverse etiologies, such as inflammatory, circulatory, or degenerative lesions. It can also be affected by tumors that are not only responsible for critical vision lossbut also for the loss of life.3 Tumors that involve the conjunctiva may be classified as benign, premalignant, or malignant.4
METHODOLOGY
This article presents a non-systematic literature review. For the survey of the literature, articles were searched for in the Scientific Electronic Library Online, Latin American and Caribbean Health Sciences Literature, and the Medical Literature Analysis and Retrieval System Online. The descriptors usedwere ocular neoplasms, carcinoma, and neoplasms of the conjunctiva. For the selection of items that served as the foundation, particularly relevant articles on the subject of this review were searched for.
LITERATURE REVIEW
EPIDEMIOLOGY AND CLASSIFICATION
Conjunctiva tumors may originate from any cell in this structure; however, the most common are of epithelial or melanocytic origin, such as squamous cell carcinoma (SCC) and melanoma, respectively.4 Tumors of the stratified conjunctival epithelium are the most commonly found primary ocular malignancy in clinical practice. They are most frequent in the region of the limbus (the transition between the sclera and the cornea).5 It is estimated that the incidence of this tumor is approximately 20 cases per one million people per year; however, a study conducted at the Ocular Oncology Unit of the Hospital Clínico Universitario de Valladolid, Spain, for 14 years, found that of a total of 314 diagnosed conjunctival tumors, 48% were melanocytic and 39.6% epithelial origin.6 Table 1 lists some of the main conjunctival tumors based on their epithelial origin.
Squamous papilloma: Squamous papilloma presents itself as an irregularly shaped pinkish exophyticlesion, with an appearance similar to a mass of highly vascularized soft tissue. Its pathogenesis may be correlated with human papilloma virus types 6, 11, 16, 18, and 33 as well as with exposure to ultraviolet rays, immunodeficiency, and tobacco smoke; in such cases, the tumor may reoccur. It has different aspects in the child, with anatomic preference in the lower fornice region, and in the adult, with main site of involvement in the bulbar eyelid. It is usually asymptomatic, with no signs of inflammation. Histologically, it is characterized by numerous papillary vascular axes covered by the acanthotic conjunctival epithelium with no signs of atypia.4,6 This tumor most typically affects adults between 20 and 39 years of age.7 8
Pseudoepitheliomatous hyperplasia and pseudocarcinomatous hyperplasia: These tumors present as a hyperplasia process that can arise from a preexisting inflammatory lesion on a pterygium or pinguecula for example. Clinically, hyperplasia presents as a fast-growing lesion that can develop as a raised mass of hyperkeratosis. Histologically, the epithelium proliferates, forming lobes, and there is an increase in mitotic activity inthe region.4,7
Actinic keratosis: It is a lesion that usually develops on a pterygium or pinguecula that is chronically inflamed. Epithelial proliferation occurs more frequently in the bulbar conjunctiva. These tumors may simulate a carcinoma. Clinically, they are well circumscribed and raised. Unlike pseudoepitheliomatous hyperplasia and pseudocarcinomatous hyperplasia, actinic keratosis exhibits slow growth. The histology of actinic keratosis may exhibit anything from slight acanthosis to severe pleomorphism and dyskeratosis, with abnormal mitosis in the region.7,9
Corneal and conjunctival intraepithelial neoplasia (CIN): It is a lesion on the epithelium of the ocular surface, with a low rate of malignancy (approximately 5% of cases). It is characterized by lesions that are gelatinous, nodular, whitish, and opaque, and it is most commonly located near the limbus. Histologically, the cells have a pleomorphic nature, with atypia to the depth of the conjunctival epithelium; the basal membrane is intact, a characteristic that helps todifferentiate between this lesion and invasive SCC. The corneal and CIN develops slowly.10,11
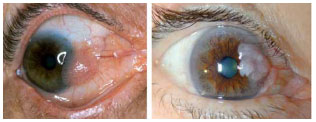
SCC: It is a conjunctival lesion in which abnormal cells are no longer limited to the epithelium of the conjunctiva, and they invade the conjunctival stroma.12 Its most common location is the limbus on the bulbar conjunctiva, and it is rarely found on the palpebral conjunctiva or isolated on the epithelium of the cornea. The lesion is either plate-shaped or in the form of a node; it is exophytic and pedunculated. Data in the literature indicate that SCC is unlikely to metastasize; however, immunocompromised patients are at an increased risk for metastasis.4,13 Histologically, the pleomorphic andhyperchromatic cells may be indistinguishable from fibroblasts, and the lesion may be confused with an amelanotic melanoma, a leiomyosarcoma, a malignant fibrous histiocytoma, or a fibrosarcoma.14
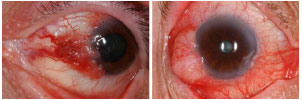
Spindle-cell carcinoma: Its incidence is low. It is aggressive and tends to penetrate the eyeball. It presents as a flat lesion. Histologically, the cells are fusiform; have hyperchromatic nuclei; and in some cases, may be difficult to differentiate from fibroblasts.
Mucoepidermoid carcinoma: As in the case of spindle-cell carcinoma, mucoepidermoid carcinoma is rare and more common in the seventh decade of life. This type of lesion is formed by mucus-secreting cells and by skin cells. Clinically, it presents as a limbal or perilimbal mass and is typically interpalpebral. It differs in some cases from SCC by the fact that the latter is more yellowish, globular, and cystic. It isimportant to make this differential diagnosis because mucoepidermoid carcinoma is more aggressive and has a greater chance of metastasizing to the eyeball and the orbit. The lesion is usually found in the conjunctival sac.4,7
DIAGNOSTIC METHODS
The technique of choice in cases in which it is necessary to collect material for diagnosis is excisional biopsy, which, in addition to providing material for histopathology (the gold standard in the diagnosis of conjunctival lesions), has therapeutic functions because the treatment of choice is surgical excision. If the excision does not have enough free borders to prevent recurrence or the development of metastases, adjuvant therapy such as chemotherapy, radiation therapy, or cryotherapy may be indicated. However, cytological examination by conjunctival scraping is easier to perform.
A study on the efficacy of cytology relative to histopathology15 found that cytology is of significant importance for differentiating between benign and malignant tumor lesions. The Papanicolaou cytological smear method is an established laboratory technique for the detection of neoplasia in tissue samples. The cytology of tissue shed from the conjunctiva and the cornea allows for an examination of the surface cell layers. These cells are derived from the layers of basal or germ cells, which undergo morphological changes in their maturation process. The abnormal maturation of cancer cells will therefore be reflected in superficial cells with anomalous features. This cytology principle allows for the examination of the tissue according to the morphology of the surface cells.15 To clarify the effectiveness of cytology by the Papanicolaou smear method relative to histopathology, a prospective study16 was conducted in the city of São Paulo, wherein the cytological sample was collected using a plastic brush, followed by fixationwith 90% ethanol on a slide. An excisional biopsy was then performed and sent to the pathology department as a double-blind examination, the results of which provided sensitivity, specificity, positive predictive value, and negative predictive value results of 85.7%, 86.7%, 75%, and 92.9%, respectively, indicating that the cytology of the conjunctival lesions is a simple procedure with a high correlation with histological monitoring. Its widespread use could increase the early diagnosis of ocular surface squamous neoplasia, with improvement in patients’ prognosis.16
Impression cytology is an alternative to conjunctival scrapings. It consists of a non-invasive method for assessing the ocular surface, including the epitheliumof the conjunctiva and the cornea. It can also ensure better quality samples. When applied on the conjunctival-corneal surface, the filter paper removes samples containing one to three layers of epithelial cells and preserves the morphological characteristics and anatomic relationships of the cells obtained. The samples are fixed, stained, and examined in the laboratory using amodified method for studying the cytology of the ocular surface. This technique aids in the understanding and guides the treatmentof disorders of the ocular surface: it enables the analysis of the degree of squamous metaplasia as well as goblet cell counts in cases of dry eye or hypovitaminosis. To assess the extent of infiltration, it is possible to use ultrasound biomicroscopy, which evaluates the deep anterior structures of the eye such as the cornea, anterior chamber, iris, and lens.17,18 For the deeper structures, there is three-dimensional optical coherence tomography, the only option capable of providing a detailed and three-dimensional assessment of the retina and the optic nerve. Even the smallest variations in these structures can bedetected.18 In unclear or difficult-to-assess cases, a computed tomography scan of the eye socket can also be performed.
TREATMENT
The treatment of epithelial tumors of the conjunctiva is based on three main pillars: destruction or removal of the neoplastic lesion via surgery and/or adjuvant treatment (procedures such as topical chemotherapy, radiotherapy, and even cryotherapy may also be indicated to reduce chances of recurrence); an early and accurate histopathological diagnosis because the clinical examination of the lesion is often inconclusive; and finally, preventing recurrences of the tumor, which may occur mainly in the first 2 years after surgery. Therefore, postoperative monitoring is necessary for at least 5 years.19
Therefore, it is necessary to perform a detailed preoperative evaluation to determine the clinical characteristics of the lesion, such as the presence or absence of bilateral, intraorbital, or intraocular invasion; to evaluate whether the tumor is diffuse or circumscribed; and to identify metastases, though these are rarely found. These characteristics help determine which surgical technique can be performed in each case. In the case of a circumscribed tumor, complete removal via excisional biopsy with minimum handling and respecting a resection border of 3–5 mm may be sufficient treatment. Bowman's capsule must be respected because in cases in which it is removed, there is a greater ease of intraocular penetration if the lesion recurs. Intraoperative assessment of the surgical borders, associated with cryotherapy, has demonstrated a lower rate of recurrence in cases of premalignant and malignant lesions. In diffuse and extensive lesions that cannot be removed completely and that do not have the minimum borders necessary, an excision must be performed to the maximum extent possible. It must be associated with an adjuvant therapy for the residual lesion using topical chemotherapeutic drugs such as mitomycin C (MMC), 5-fluorouracil, or interferon alpha 2 beta, and with radiotherapy.4 During surgery and even postoperatively, 1% toluidine blue is used to outline the lesion borders and even to detect possible lesion recurrence as early as possible. Its efficacy was demonstrated in a prospective study on the use of 1% toluidine blue eye drops for the diagnosis of squamous cell cancers of the ocular surface.21 The 1% toluidine blue test resulted in 90% efficacy for positive staining used in the detection of premalignant lesions and 100% efficacy in the case of malignant lesions. These lesions were subsequently proven using a cytological study, and only 1 out of 47 patients studied presented a false positive. Therefore, 1% toluidine blue is a simple procedure that is accessible to all ophthalmologists. It assists in early diagnosis and the effective treatment of squamous cell cancer of the ocular surface.20
Topical chemotherapy has advantages over surgery in thatit enables treatment of the entire ocular surface and does not require surgical borders. In addition, its use in neoadjuvant therapy prevents possible surgical complications. In cases in which thepatient does not accept the surgical procedure, it has been used as the primary treatment.21,22. MMC, for example, acts by inhibiting DNA synthesis, and is more effective in rapidly dividing cells. This agent has been used as an adjuvant for CINand SCC excision preoperatively, intraoperatively, and postoperatively. However, other studies have recommended its use as a primary therapy for CIN/SCC, evenin cases of recurrent disease.11
A prospective study conductedto evaluate safety, efficacy, and tumor recurrence after the use of MMC in the treatment of primary and recurrent CIN and of recurrent SCC studied two groups of patients. G1 (CIN) was treated with 0.02% MMC eye drops 4 times daily for 28 days, and G2 (recurrent SCC) was treated with 0.02% MMC eye drops 4 times daily for 21 to 28 days. In the results, eight patients experienced complete regression of the lesion with negative cytology 1 month after the end of treatment, and one patient exhibited partial regression of the lesion and underwent excisional biopsy, which was negative for dysplasia. Of the patients with SCC, 1 experienced complete regression of the lesion and 2 experienced partial regression, with negative pathologic results. Thus, it is suggested that the use of topical MMC is a safe and effective option as a primary single treatment for recurrent or primary CIN and for recurrent SCC; however, in most of these cases, surgery was still indicated.22 In cases in whichintraocular invasion or enucleation is indicated as well as when previous orbital invasion exists, earlier excision with eyelid preservation is indicated, provided they are not affected.
Adverse effects of MMC: According to a study on the effectiveness ofmitomycin,24 adverse effects found in the patients during treatment with MMC were conjunctival hyperemia, lacrimation, sensations of a foreign body, and secretion. On biomicroscopy, the most frequent abnormalities were diffuse conjunctival hyperemia, punctate keratitis, mucus secretion, and epithelial defects. The suspension of chemotherapy was sufficient for the regression of ocular changes, without the need for medication.
Alternatively, interferon alpha 2 beta has been used as of late. It has fewer side effects and has therefore been better tolerated by patients. Subconjunctival administration of interferon alpha 2 beta has side effects with symptoms similar to a flu-like syndrome (fever, myalgia, fatigue) and mild hepatic disorders. When used topically, the most commonly reported adverse effects are conjunctival hyperemia and follicular conjunctivitis. A combination of administration routes (topical and subconjunctival) has been used for the primary treatment of conjunctival intraepithelial carcinoma and its recurrence. In addition, with respect to the chemotherapeutic treatment of eye tumors, there is the possibility of installing a radioactive plate in a peritumoral intraocular region, a kind of therapy known as brachytherapy. Ruthenium-106, rhodium-106, iodine-125, palladium-103, gold-198, cobalt-60, and iridium-192 can be used as radionuclides in the treatment of ocular tumors.23,25
REFERENCES
1. Hospital das Clínicas (São Paulo), organizador. Clínica Oftalmológica. Anatomia e fisiologia do olho: anatomia e fisiologia [Internet]. Cap. 1 [cited 2017 Feb 20]. Available from: https://www.passeidireto.com/arquivo/17566215/oftalmologia ---apostila-usp.
2. Junqueira LC, Carneiro J. Histologia básica: texto e atlas. 10a ed. Rio de Janeiro: Guanabara Koogan; 2004.
3. Barsotti V, Aguiar GMF, Abatti REM, Boldrini L, Kumagai KM, Freitas JAH. Carcinoma espinocelular de conjuntiva. Rev Fac Cienc Med Sorocaba. 2007;9:23-6.
4. Saornil MA, Becerra E, Méndez MC, Blanco G. Tumores de la conjuntiva. Arch Soc Esp Oftalmol. 2009;84:7-22. https://doi.org/10.4321/S0365-66912009000100003
5. Crawford JB. Conjunctival tumors. In: Tasman W, Jaeger EA, editors. Duane's clinical ophthalmology.Philadelphia: Lippincott; 1995. v. 4, p. 1-10.
6. Yanoff M, Duker JS. Oftalmologia. In: Goldstein MH, editor. Córnea e doenças da superfície ocular. 3a ed. Rio de Janeiro: Elsevier; 2011. p. 241.
7. Pereira APF. Lesões não melanocíticas da conjuntiva: benigna, pré-malígna e maligna [dissertação]. Coimbra: Faculdade de Medicina, Universidade de Coimbra; 2010.
8. Font RL, Del Valle M, Aveda-o J, Longo M, Boniuk M. Primary adenoid cystic carcinoma of the conjunctiva arising from the accessory lacrimal glands: a clinicopathologic study of three cases. Cornea. 2008;27:494-7. https://doi.org/10.1097/ICO.0b013e318162a907
9. Dutton JJ, Fowler AM, Proia AD. Dermoid cyst of conjunctival origin. Ophthal Plast Reconstr Surg. 2006;22:137-9. https://doi.org/10.1097/01.iop.0000199251.29558.0b
10. Vivó JM, Hernández NH, Sanz AM. Neoplasia intraepithelial córneo-conjuntival. A propósito de um caso. Rev Med Electron. 2012;34:490-5.
11. Figueirêdo RS, Figueirêdo ES. Uso de mitomicina C em neoplasia intra-epitelial córneo-conjuntival: modalidades de abordagem –relatos de caso. Arq Bras Oftalmol. 2006;69:407-11. https://doi.org/10.1590/S0004-27492006000300023
12. Ros SI. Tumores de conjuntiva (presentacion de caso clinico). Rev Med Costa Rica Centroamérica. 2007;580:133-7.
13. Perdigão FB, Pierre-Filho PTP, Natalino RJM, Caldato R, Torigoe M, Cintra ML. Conjunctival keratoacanthoma. Rev Hosp Clín Fac Med São Paulo. 2004;59:135-7. https://doi.org/10.1590/S0041-87812004000300008
14. Huntington AC, Langloss JM, Hidayat AA. Spindle cell carcinoma of the conjunctiva. An immunohistochemical and ultrastructuralstudy of six cases. Ophthalmology. 1990;97:711-7. https://doi.org/10.1016/S0161-6420(90)32526-5
15. Lima CGMG, Veloso JCB, Tavares AD, Jungman P, Vasconcelos AA. Método citológico e histopatológico no diagnóstico das lesões da conjuntiva: estudo comparativo. Arq Bras Oftalmol. 2005;68:623-6. https://doi.org/10.1590/S0004-27492005000500009
16. Kayat KV, Dantas PEC, Felberg S, Galvão MA, Saieg MA. Exfoliative cytology in the diagnosis of ocular surface squamous neoplasms. Cornea. 2017;36:127-30 https://doi.org/10.1097/ICO.0000000000001027
17. Michalany J. Técnica histológica em anatomia patológica: com instruções para o cirurgião, enfermeira e citotécnico. São Paulo: EPU; 1981. p.182-6.
18. Vide Oftalmologia. O que é OCT? [Internet]. São Paulo; 2013 [cited 2013 Feb 20]. Available from: http://videoftalmologia.com.br/?p=94
19. Barros JN, Mascaro VLDM, Gomes JAP, Freitas D, Lima ALH. Citologia de impressão da superfície ocular: técnica de exame e de coloração. Arq Bras Oftalmol. 2001;64:127-31. https://doi.org/10.1590/S0004-27492001000200007
20. Cema Hospital. Biomicroscopia Ultrassônica (UBM). São Paulo; 2017 [cited 2017 Feb 20]. Available from: http://www.cemahospital.com.br/biomicroscopia-ultrassonica-ubm
21. Romero IL, Ballalai PL. Uso do colírio azul de toluidina a 1% no diagnóstico das neoplasias de células escamosas da superfície ocular. In: 19o Congresso Brasileiro de Prevenção da Cegueira e Reabilitação Visual; 2010; Salvador. Arq Bras Oftalmol. 2010;73:14. TL 018.
22. Tonietto AP, Magnani AC, Mársico J, Mason EM, Fortes Filho JB. Carcinoma de células escamosas da conjuntiva: aspectos clínicos e histopatológicos em cinco pacientes. Rev Bras Oftalmol. 2005;64:140-5.
23. Huerva V, Ascaso FJ. Conjunctival intraepithelial neoplasia – Clinical presentation, diagnosis and treatment possibilities. In: Srivastava S, editor. Intraepithelial neoplasia [Internet]. Rijeka: InTech; 2012 [cited 2017 Feb 20]. Available from: http://www.intechopen.com/books/intraepithelial-neoplasia/conjunctivalintraepithelial-neoplasia-clinical-presentation-diagnosis-and-treatment-possibilities. https://doi.org/10.5772/38878
24. Ballalai PL, Gomes JAP, Santos MS, Freitas D, Erwenne CM, Rigueiro M. Uso de mitomicina C tópico no tratamento da neoplasia intra-epitelial córneo-conjuntival e carcinoma espinocelular conjuntival: resultados preliminares. Arq Bras Oftalmol. 2003;66:559-62. http://dx.doi.org/10.1590/sooo4-274920030000500003
25. Mattos FR, Rostelato MECM, Zeituni CA, Souza CD, Peleias Júnior FS, Benega MAG, et al. Desenvolvimento de um dispositivo de suporte de sementes de braquiterapia para irradiação. In: 14o Congresso da Sociedade Brasileira de Radioterapia; 2012; Curitiba. SãoPaulo: Ipen; 2014. 3 p.



Funding source:None
Conflicts of interest:None
Received on:
February 15, 2017.
Accepted on:
March 10, 2017.